Company Overview
GeneDx Holdings Corp. (NASDAQ:WGS) specializes in delivering precise medical diagnostic results by leveraging its genomic and data science expertise, particularly its extensive rare disease dataset. This allows the company to interpret large volumes of clinical data at scale. GeneDx focuses on pediatric care, using whole exome and whole genome sequencing tests to identify rare diseases and deliver more definitive diagnoses. Additionally, it licenses its rare disease dataset and expertise to accelerate drug discovery.
Founded in 2000 by scientists from the National Institutes of Health (NIH), GeneDx was initially established to make genetic testing accessible for rare disease patients. It was incorporated in July 2020 as CM Life Sciences, Inc. In July 2021, CM Life Sciences merged with Sema4, which in April 2022 acquired GeneDx. In January 2023, Sema4 Holdings was renamed GeneDx Holdings Corp.
A Short Primer
Genes, composed of DNA, are the fundamental building blocks of life and heredity. The entire set of genes in an individual is known as a genome. Within the human genome, approximately 19,900 genes are involved in protein coding. These protein-coding regions, called exons, are collectively known as the exome. The exome makes up about 2% of the genome, but encodes most disease-related gene variants. Exome sequencing studies these regions, while genome sequencing examines the entire genome. Genetic testing includes molecular, chromosomal, gene expression, and biochemical tests.
Exome and Genome Testing as a Growth Driver
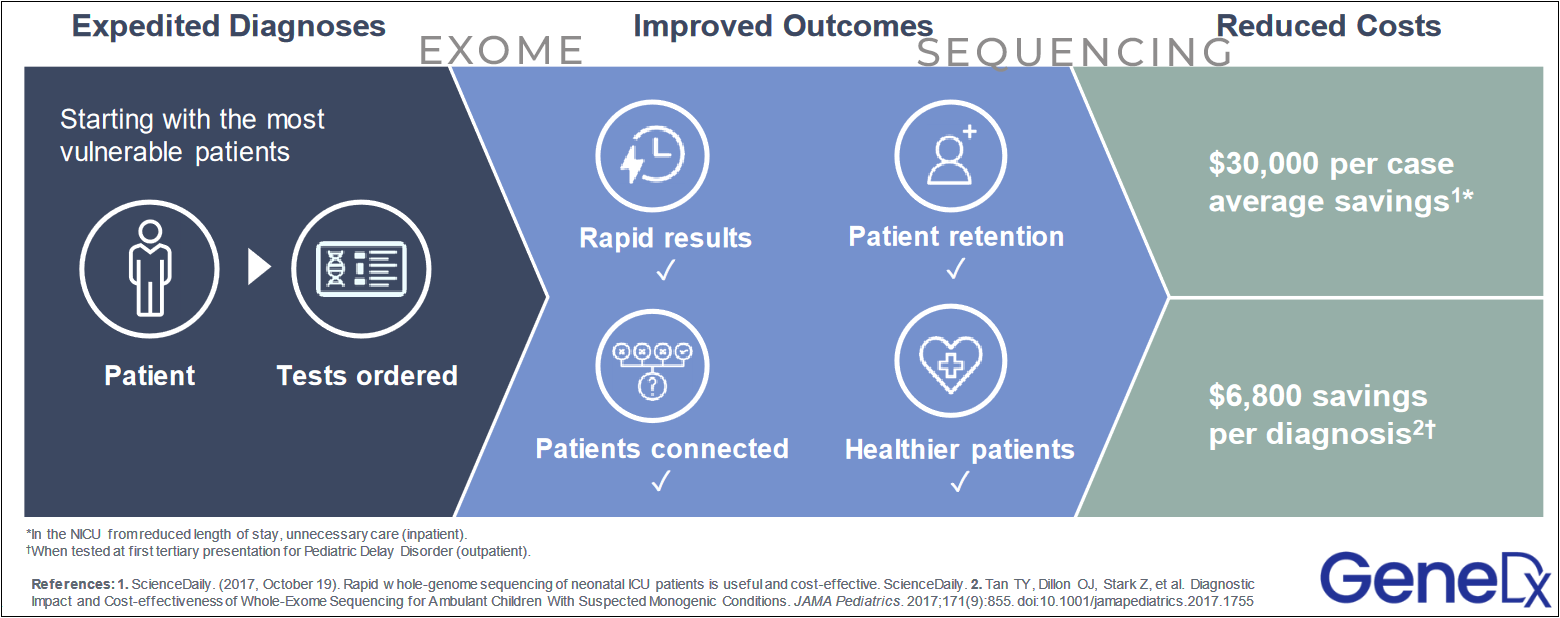
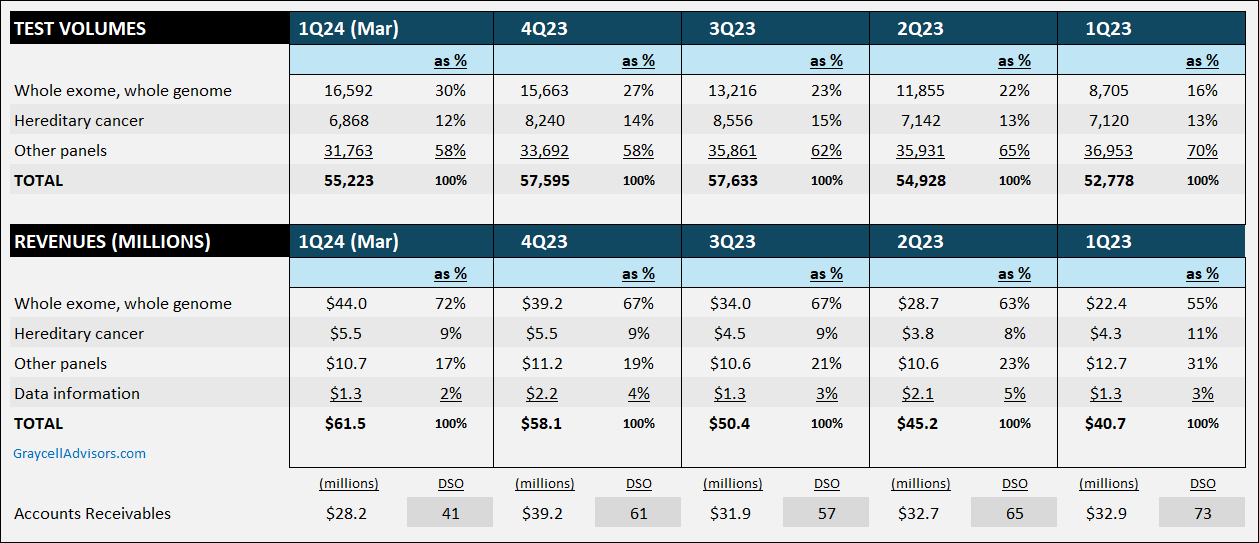
GeneDx offers molecular tests, specializing in Whole Exome Sequencing (WES) and Whole Genome Sequencing (WGS). This segment, accounting for 70% of revenues in the first quarter of 2024, is key to achieving profitability in 2025 and presents a primary reason for investor interest. Whole exome testing is highly effective in early disease identification, enabling timely intervention and reducing healthcare costs from misdiagnoses.
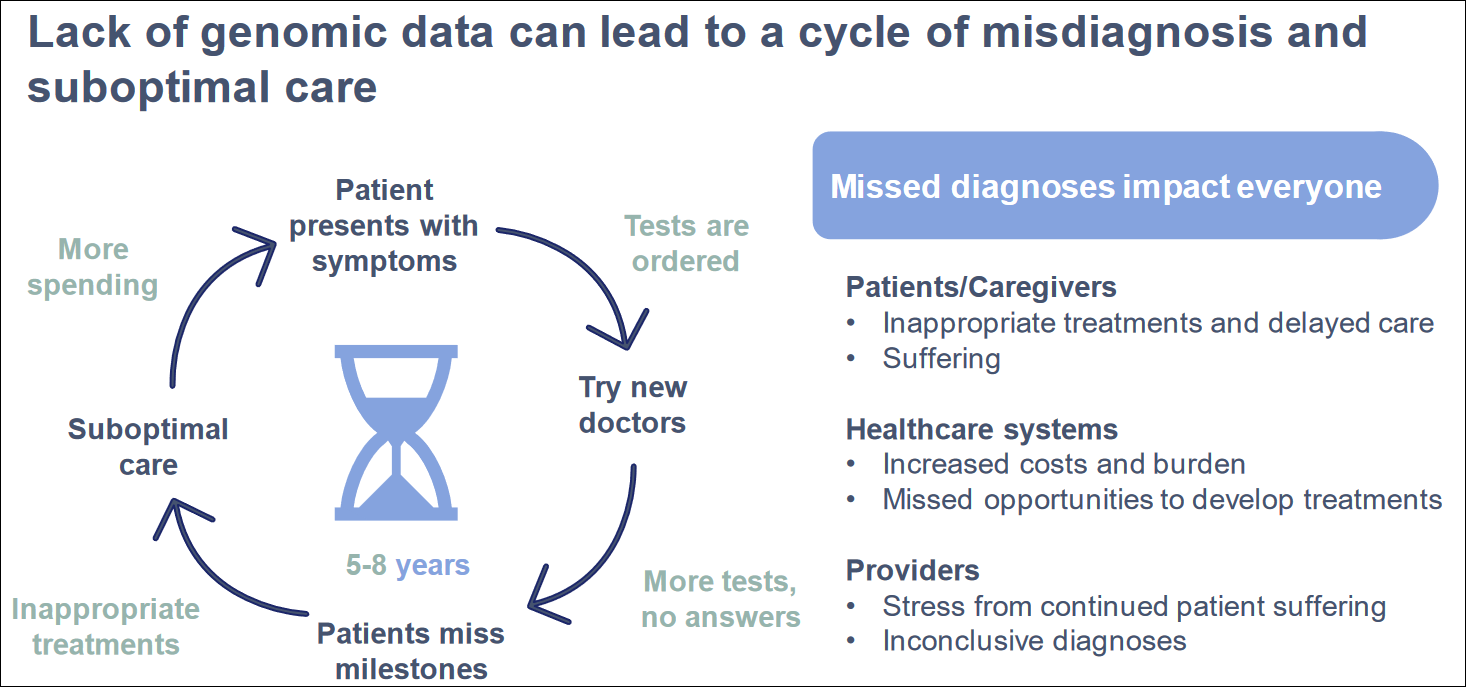
Cost of Misdiagnosis
The whole exome tests offer a higher probability of identifying genetic diagnoses, compared to other gene tests, which may not provide definitive results. Many genes can be involved in major diseases, and a whole exome or genome test is more effective. For instance, the American Epilepsy Society's 2023 Annual Meeting reported that 768 genes are related to epilepsy seizures, but commercial gene panel testing only covers 43% of these. Exome sequencing tests all related genes. GeneDx targets the pediatric care market, demonstrating the value of early genome testing.
Studies like the SeqFirst project with the University of Washington have shown the effectiveness of rapid whole genome sequencing (rWGS) in diagnosing critically ill infants in neonatal intensive care units.
GeneDx is supporting whole genome sequencing and interpretation services for the GUARDIAN study sponsored by Columbia University, by providing screening for up to 100,000 newborns for more than 450 rare genetic conditions not currently included in standard newborn screening. Data from the study of 5000 infants has shown an over 4% positive rate for rare diseases.
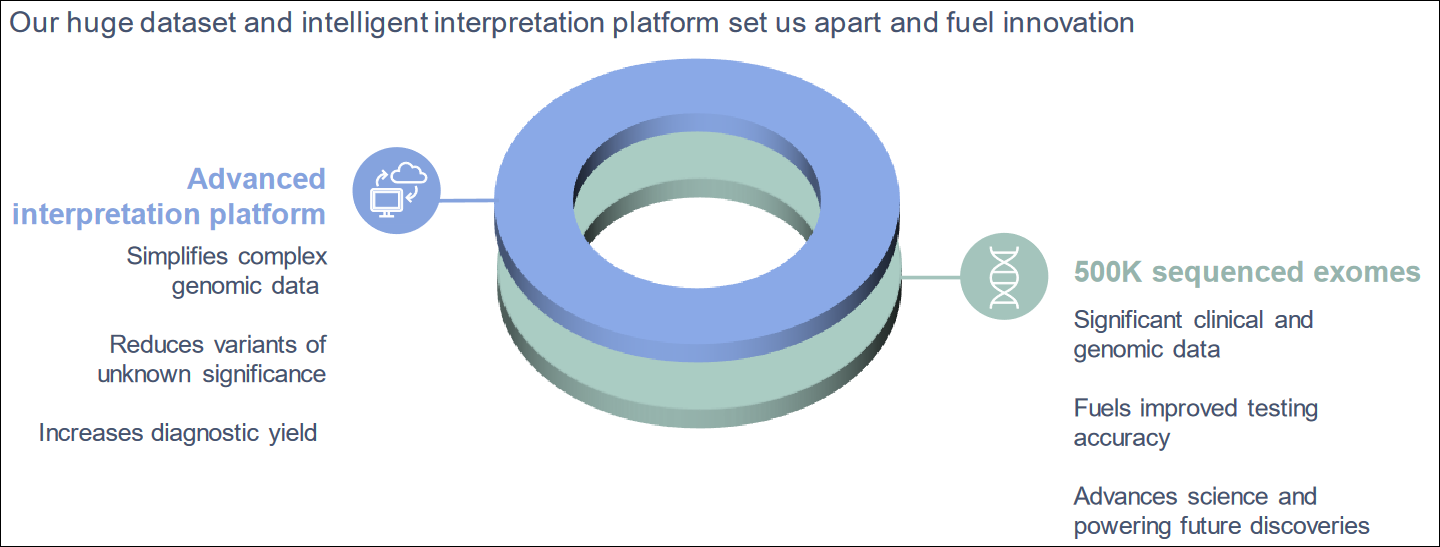
A Gene Data Interpreting Platform
GeneDx's extensive rare disease dataset, one of the largest, is a significant competitive advantage. With over 600,000 sequenced exomes and genomes, the company leverages its data science expertise to deliver genomic diagnostic data at scale. As more genomes and exomes are sequenced, the data interpretation platform continues to mature, enhancing precision in diagnosis and insights. Such a critical mass of scale and platform intelligence can become a major competitive edge that ramps up revenues and builds market share as gene diagnostic testing grows.
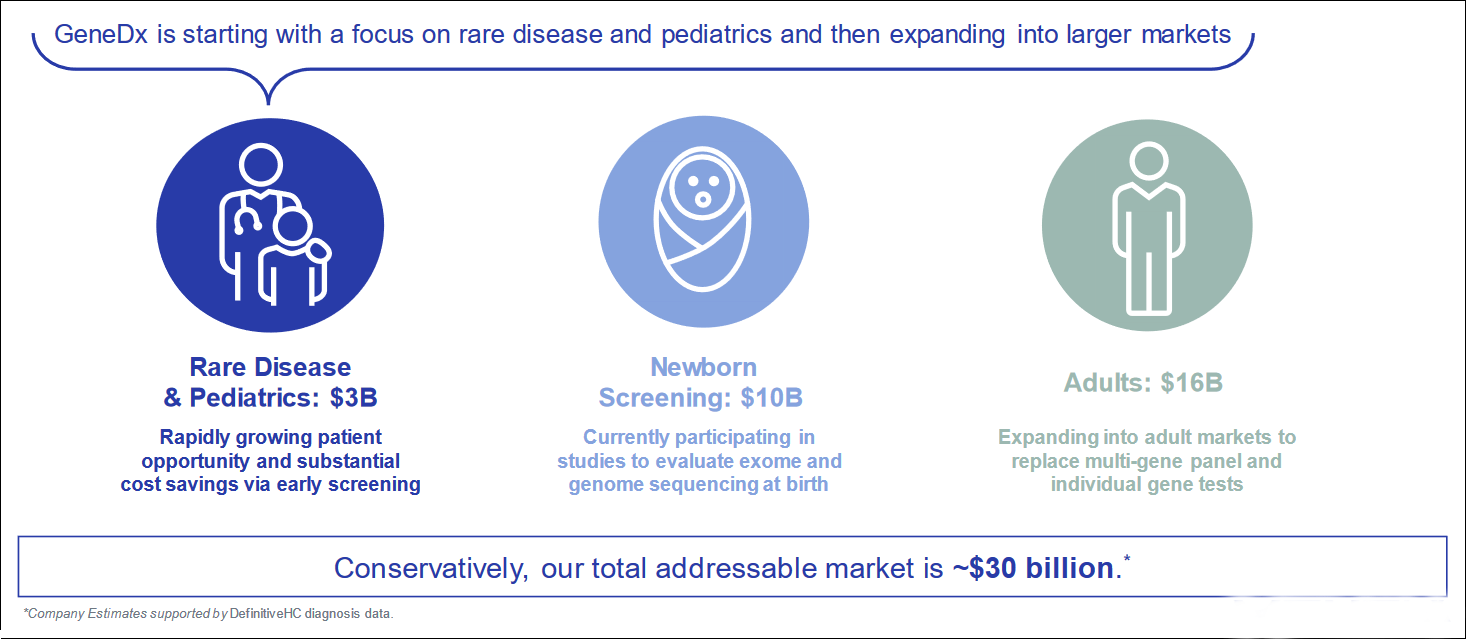
Growing Market Acceptance & Opportunity
The shift towards cost-effective diagnostics creates opportunities for exome and genome testing to become a standard for diagnosing genetic diseases.
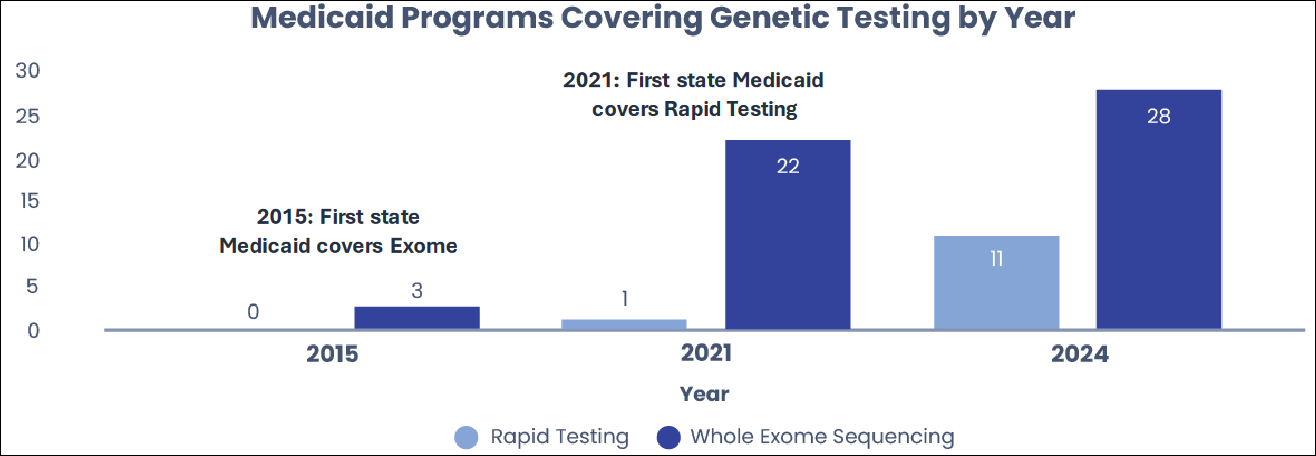
Healthcare spending is determined by payor coverage, which is expanding in the case of whole genetic sequencing. GeneDx's services are contracted with major commercial payors and Medicaid, and the state coverage is expanding. As of now, there are 28 states which cover exome sequencing and 11 states cover rapid genome sequencing.
Financials
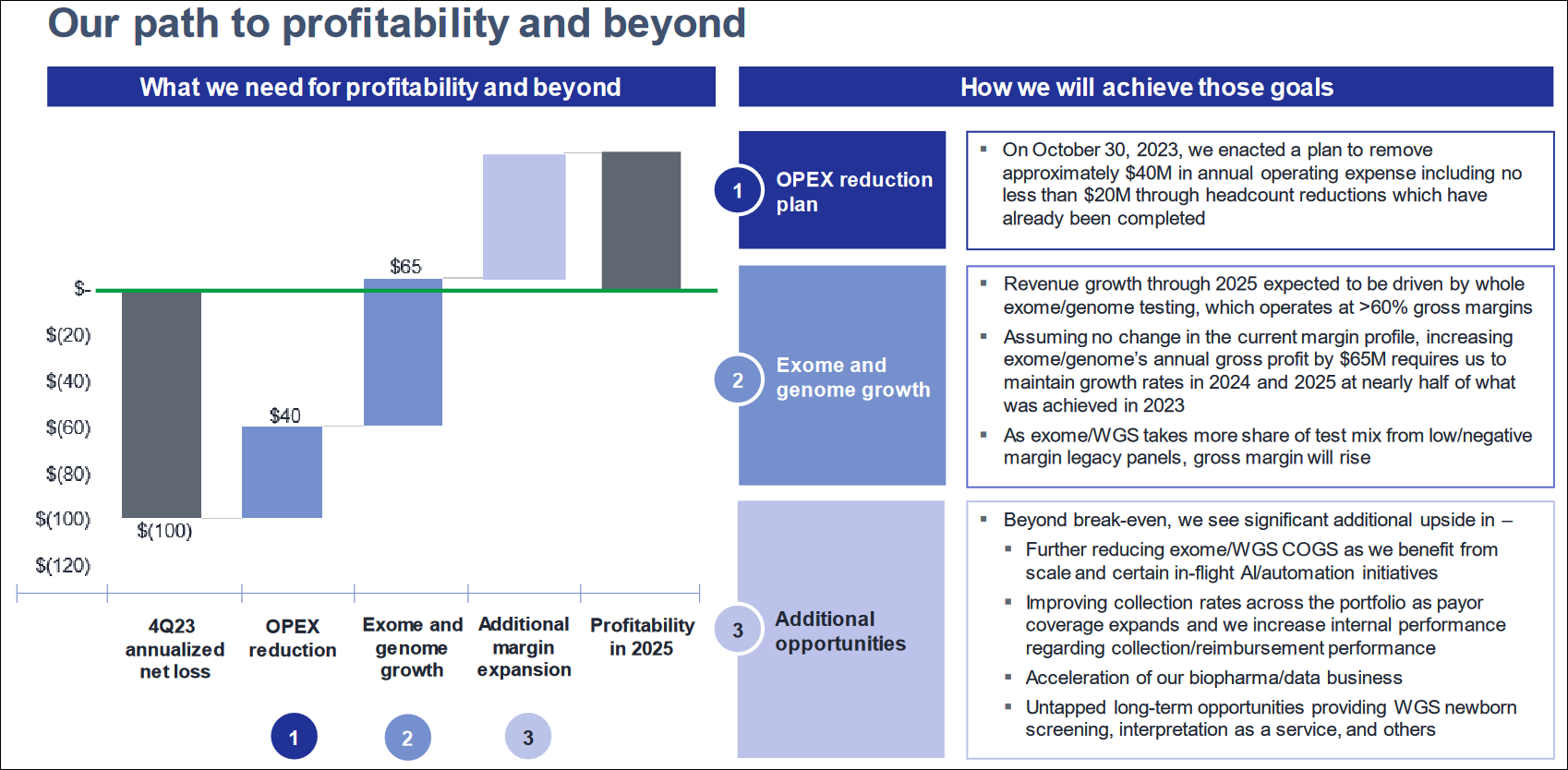
GeneDx's revenue growth reflects the increasing acceptance of genome and exome testing, which is a higher-margin business. In the first quarter of 2024, total revenues grew 51% compared to the previous year, with whole exome and genome revenues surging 96%. Gross margin improved to 61% from 56%. The company reported an operating loss of $13.7 million, down significantly from $60 million the previous year. Cash burn improved significantly, with net cash used in operations declining to $16.4 million from $56 million a year ago.
As of March 2024, GeneDx had $114 million in cash and equivalents. The company also has a $75 million term loan facility with Perceptive Advisors, with $50 million utilized and $25 million available upon meeting certain conditions. In April 2024, the company filed a supplement to its shelf registration allowing it to sell up to $75 million in shares from time to time as an at-the-market offering.
Long-term debt was $52.3 million at the end of the first quarter, nearly all of it to mature in 2028.
Management
The management team is led by Katherine Stueland, who in June 2021 became President and CEO of GeneDx, a company founded by NIH scientists. She became co-CEO of the combined company when it was acquired by Sema4, and eventually its sole CEO, playing a defining role since in the rebuilding of the new GeneDx.
Earlier, Ms. Stueland was the Chief Commercial Officer of Invitae, a medical genetics company. GeneDx's management team includes experienced professionals from the genetic testing industry. The team is focused on driving the company to profitability in 2025, emphasizing growth in exome and genome testing, reimbursement rates, and cash burn reduction. The laser focus is important in a highly competitive medical diagnostics market with companies like Natera (NTRA), NeoGenomics (NEO), and now Lab Corp. with its acquisition of Invitae.
The company must maintain growth, improve gross margin, and reduce operating expenses, in its quest for profitability, and the path it has laid out reflects that.
Risks
GeneDx faces several risks, including dependency on payor coverage and reimbursement rates. The company's principal customer groups include the healthcare professionals, who bill the third-party insurance providers (commercial or government) and represent about 72% of the revenue billing, and institutional clients, like hospitals and laboratories, representing over 27%. Any changes in payor guidelines or disputes can significantly impact the company's liquidity and business. Exposure to a single-payor, like a major health insurance company, can be high even though it represents multiple claims and plans, and any disagreement can have a magnified impact.
In December 2022, GeneDx settled with a payor for billing issues that resulted in overpayments to the legacy Sema4 business, which requires GeneDx to pay $42 million to the payor in a series of payments through June 2026.
With an accounts receivable concentration with payors, managing collection is vital, and the company's days of sales outstanding have been trending lower, standing at 41 days in the March quarter, compared to 73 days a year ago.
Conclusion
GeneDx is a turnaround company at a pivotal juncture, leveraging its advanced genomic testing capabilities to capitalize on the growing demand for precise medical diagnostics. With a strong foundation built on its extensive rare disease data set and expertise in whole exome and genome sequencing, GeneDx is strategically positioned to lead the market in pediatric care and beyond. The company's focus on early and accurate diagnosis not only benefits patient outcomes but also presents a compelling value proposition by reducing systemic healthcare costs associated with misdiagnosed treatments.
The management team, led by experienced professionals from the genetic testing industry, is committed to steering GeneDx toward profitability in 2025. This goal is supported by a clear strategy to expand exome and genome testing, enhance reimbursement rates, and reduce cash burn. As healthcare trends increasingly favor cost-effective and accurate diagnostic solutions, GeneDx's model aligns well with market needs, offering significant growth potential. An investment in GeneDx hinges on the management's ability to execute this strategic plan effectively, making it a promising candidate for those looking to invest in the future of genomic diagnostics in pediatric care.
A more favorable macro backdrop with rate cuts as the year progresses, the defensive nature of healthcare spending in a slower growth environment, and the strengths and positioning of GeneDx, can create an opportunity for the share price to advance to above $50 over the next 6 to 12 months.
GeneDx is part of the Prudent Healthcare and Prudent Small Cap model portfolios for subscribers. The positions can be closed at any time and our company opinion can change without public notification.