Company Overview
Nektar Therapeutics (NKTR) is an immunotherapy company focused on leveraging the immune system to address disease areas such as autoimmune and oncology. Using its immunology expertise, the company is designing novel immunomodulatory drugs that can enhance, regulate, or suppress immune activity to achieve targeted and meaningful clinical benefits in treating the disease. Earlier this year, the company's Phase 2 results for Atopic Dermatitis, an inflammatory skin condition characterized by an overactive immune response, met its endpoints, boosting confidence in the approach and platform potential.
Balancing and Leveraging the Immune System
Nektar is focused on novel approaches to correct the imbalance of regulatory T-cells to target the resulting dysfunction, thus restoring the mechanisms of self-tolerance to achieve a state of immune homeostasis.
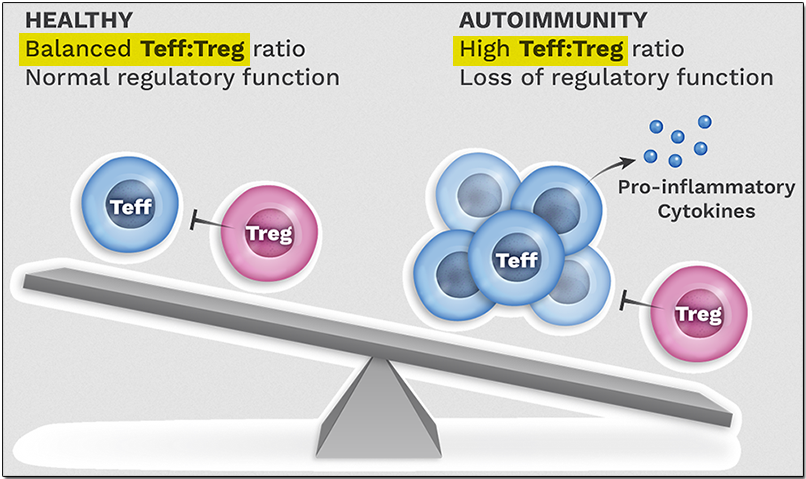
The immune system maintains a state of tolerance or balance in a process called immune homeostasis, protecting the body by fighting pathogens while preventing the formation of pathogenic T-cells that can cause autoimmune reactions by mistakenly attacking the body's own cells.
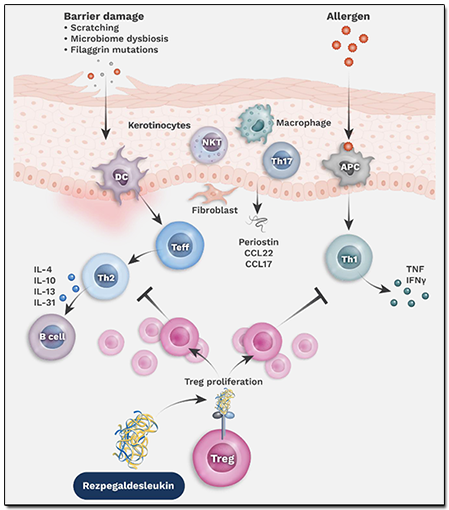
It is a dynamic equilibrium involving complex mechanisms that allow the immune system's regulatory T cells (Tregs) to direct the effector T cells (Teffs) in responding sufficiently to threats, while preventing the Teffs from becoming overactive. This dynamic is shown in the illustration below.
A higher number of Teffs can tilt the immune system balance towards inflammation. Expanding the Treg count can restore the immunoregulatory balance.
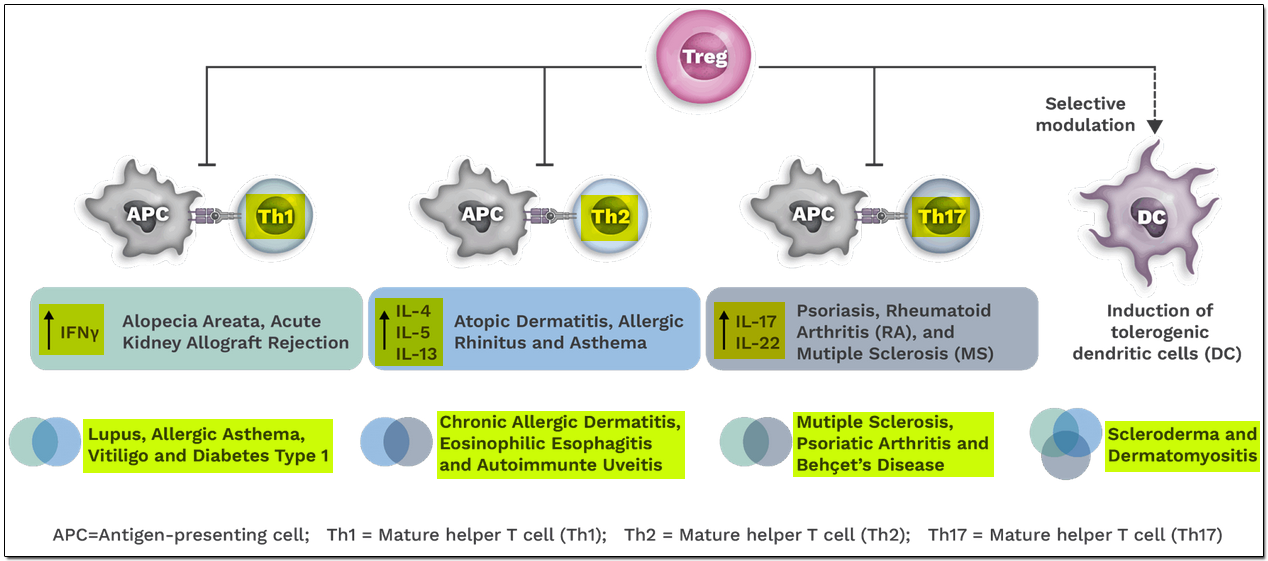
Since Tregs modulate the immune system upstream of the pro-inflammatory cytokine pathways that drive the Th1, Th2, and Th17-mediated inflammation, multiple cytokine pathways can be modulated by managing the functionality of Tregs.
The slide below is quite informative on using the Treg approach to address disease areas and also shows:
Pipeline
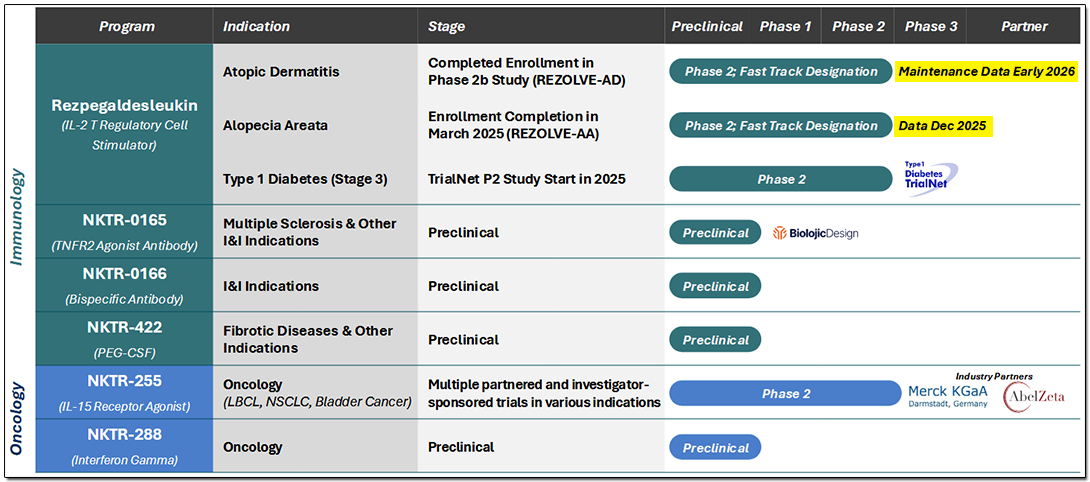
Nektar believes that its deep understanding of immune science will contribute towards developing innovative and novel drug candidates, which are immunomodulatory agents that regulate T cells by using the cytokine pathways to target immune imbalances in conditions associated with autoimmune and chronic inflammation. It is this expertise in modulating the immune system that the company is attempting to leverage in its pipeline, which covers autoimmune diseases and oncology.
Autoimmune
Autoimmune disorders and inflammatory diseases are caused by an imbalance in the body’s immune system. The company's most advanced drug candidate is Rezpegaldesleukin, also referred to as Rezpeg, which is a potential first-in-class resolution T cell mechanism targeting the underlying immune system imbalance. It has been designed to closely mimic the mechanism by which Tregs work in our body to resolve inflammation. Its constructs emulate human biology very closely through interleukin-2 (IL-2) agonism and a validated chemistry approach. The biologic targets IL-2 receptors located on Tregs to proliferate and increase their functionality for controlling overactive or proliferating Teffs, and thus correct the imbalance.
Due to its mechanism of action, Rezpegaldesleukin is a platform drug being evaluated for various inflammatory diseases.
Rezpeg is in a trial for the treatment of atopic dermatitis [AD] disorder, also known as eczema, a chronic inflammatory skin condition. The investigational therapy received a Fast Track designation earlier this year for treatment of adult and pediatric patients of 12 years and above whose disease is not sufficiently managed with topical therapies or when such therapies are inadvisable. Recent Phase 2b results met primary and secondary endpoints and are discussed further below.
Rezpegaldesleukin is also in Phase 2 trials as a treatment for alopecia areata [AA], where inflammation around the hair bulb causes a loss of immunity around the hair follicle, resulting in its loss. By increasing the number and functionality of regulatory T cells, this investigational therapy aims to reduce the local inflammation and restore immune privilege. In July 2025, the FDA granted a Fast Track designation for the treatment of severe and higher-grade alopecia areata in adults and pediatric patients 12 years of age and older. Trial data readout is expected in December 2025.
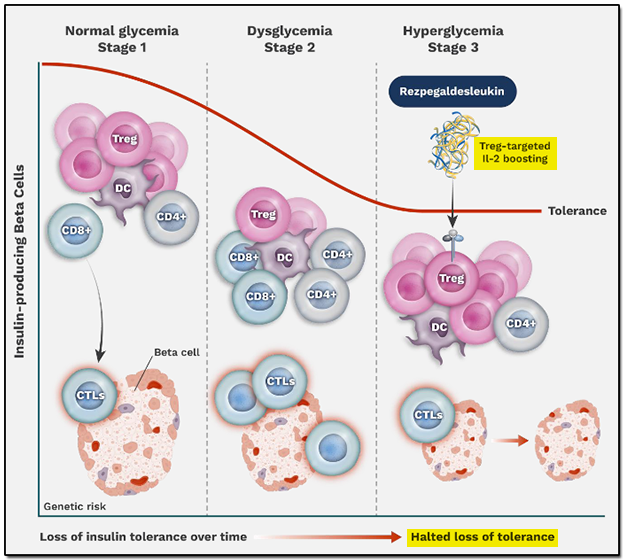
Rezpegaldesleukin is also being tested as a treatment for Type I diabetes mellitus, commonly known as diabetes, where the pancreatic cells producing insulin are attacked by the body's T cells. Rezpegaldesleukin contributes to an increase in the number and functionality of Tregs, which act upstream of the attacking T cells, thus bringing about an immunobalance that can slow the progression of new-onset Type 1 diabetes. The investigational therapy started its Phase 2 trials earlier in 2025, and the company has partnered with TrialNet, which is funding an investigator-sponsored Phase II clinical trial.
The company has a few other programs in early stage, including a preclinical investigational candidate, NKTR-0165, which is a tumor necrosis factor (TNF) receptor type II (TNFR2) agonist, which can be potentially developed for treatment of autoimmune diseases, like multiple sclerosis and ulcerative colitis.
Oncology
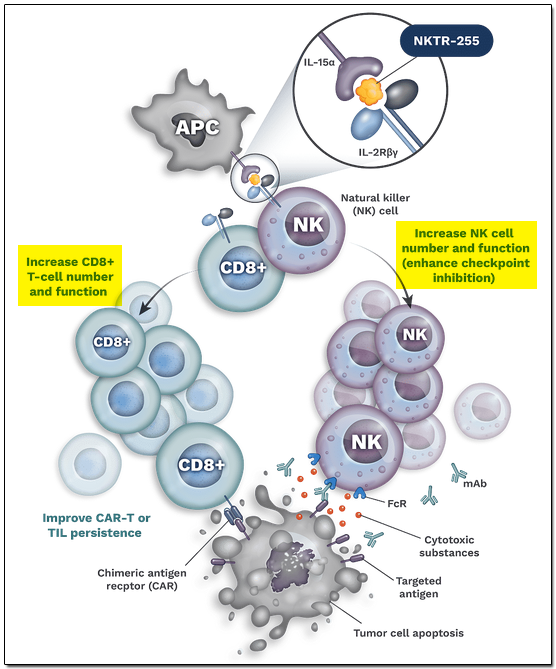
The company is focusing on targeting pathways that stimulate and sustain the immune system to fight cancer. NKTR-255 is a trial drug designed to optimally activate the interleukin-15 (IL-15) pathway to increase survival and proliferation of a variety of cells, including cancer-fighting natural killer (NK) cells, tumor-infiltrating lymphocytes (TILs), and CAR-T cells, thus amplifying and prolonging the anti-tumor response. NKTR-255 is being investigated in various indications with multiple partners, including Merck (Germany), and trial updates are expected in 2026.
Phase 2b Study REZOLVE-AD
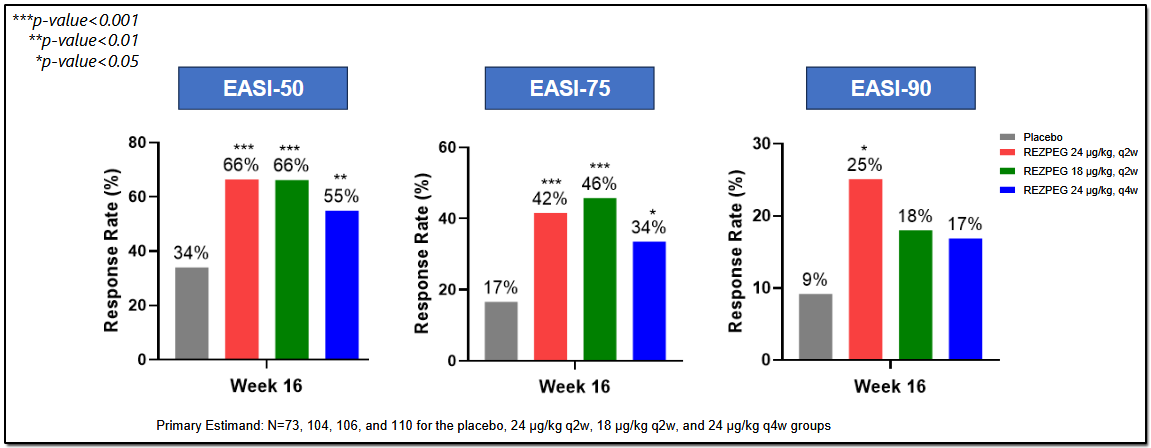
In June 2025, Nektar announced the Phase 2b trial data of its REZOLVE-AD study for atopic dermatitis.
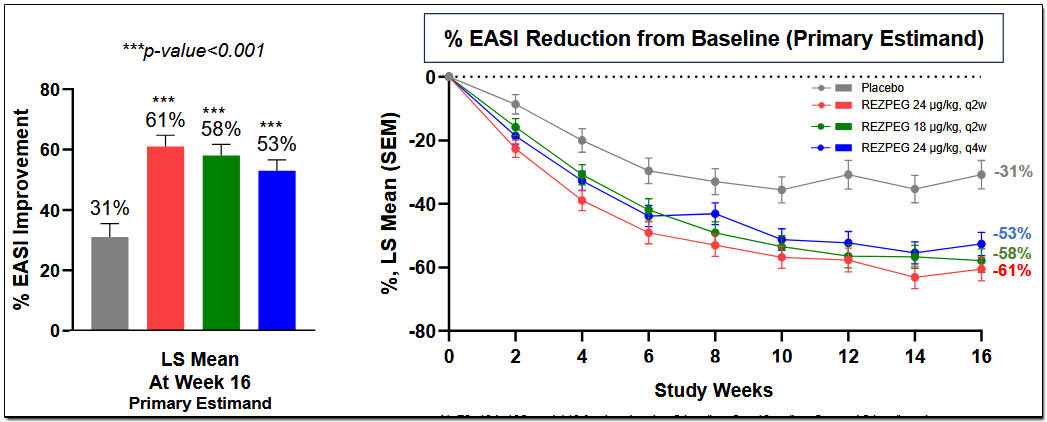
The 16-week data confirmed Rezpeg as a first-in-class regulatory mechanism of T cells in AD, with a sixfold increase in Tregs and reduction in multiple AD biomarkers. All three dose arms met their primary endpoint of mean improvement in Eczema Area and Severity Score [EASI]. The study is being conducted in 393 patients with moderate-to-severe AD.
All doses met multiple secondary endpoints, with the highest dose meeting all key secondary endpoints, including statistical significance on EASI-90, which is the percent of patients who achieved ≥ 90% reduction in EASI from baseline.
The associated press release with additional details can be viewed here. The end of the Phase 2 meeting with the FDA is expected by the end of 2025, and based on feedback, Phase 3 is expected to begin in 2026.
Additional data will be released during the first quarter of 2026 for the 52-week maintenance and escape arm from the RESOLVE-AD study, which will provide insights into the effectiveness of continued treatment with Rezpeg in patients who established an EASI-50 response at the end of 16 weeks. The data released so far has yet to see a plateau in the efficacy response of Rezpeg, and it will be interesting to learn its further evolution.
Data from a 52-week off-study treatment from Phase 2b REZOLVE-AD will be available in early 2027, which will reflect on the durability of the treatment.
Market Opportunity
The company estimates the market size for atopic dermatitis to be a multi-billion-dollar opportunity.
There are approximately 15 million adults with moderate-to-severe AD in the U.S. and about 110 million globally, as per the National Eczema Association and the Eczema Council. Although there are multiple drugs for AD, the company's research has shown a high unmet need for a novel mechanism that induces deeper and potentially therapy-free remission and offers dosing schedules without rebound effects. The current market leader is Dupixent, from Regeneron Pharmaceuticals and Sanofi, with annual sales of over $10 billion. However, the company estimates that 50% of patients fail on therapy, as per research in the New England Journal of Medicine and Evaluate Pharma. Current therapies can be limited by efficacy, side effects, dose inflexibility, and disease control over the long term.
Rezpeg is also being tested in a different dermatological setting in patients with alopecia areata, where Phase 2 results are expected in December 2025. There are approximately 7 million people in the U.S. who have or will develop AA. Over 1 million suffer from severe-to-very-severe disease, as per the National Alopecia Areata Foundation, with limited therapy options beyond a JAK inhibitor. If successful, Rezpeg can be the first biologic in the setting of alopecia areata, representing an additional $1 billion market opportunity.
Risks
The key risk for Nektar Therapeutics is similar to the one for any biotech - data disappointment.
The data have been quite strong on safety and efficacy in Phase 2 trials, which diminishes the potential for a major variance. Maintenance and escape arm Phase 2 data is still due, and Phase 3 has to be accomplished. In addition, the end-of-Phase 2 meeting with the FDA is expected by next month, and based on the agency's feedback, it will impact the design and timing of Phase 3 start.
Later in December, data for Alopecia Areata will be released, which will be for a second indication and significantly contribute towards validating the platform potential of Rezpeg for multiple treatments.
The company estimates ending 2025 with about $240 million in cash and investments, sufficient for operations till the second quarter of 2027.
Conclusion
Rezpeg incorporates a novel mechanism of action that has proven thus far to be safe and efficacious. It represents not just a single therapy but a building platform for multiple potential therapies addressing major multi-billion dollar markets.
There are a number of upcoming catalysts. In December, the topline results from the Phase IIb REZOLVE-AA study in patients with alopecia areata will be released, along with the outcome of the FDA end-of-Phase 2 meeting. In the first quarter of 2026, additional data from the atopic dermatitis Phase IIb REZOLVE-AD study will be released. Updates next year are also expected from the Phase 2 diabetes trial being conducted by TrialNet. Also, the Phase 3 start for AD and AA trials, if the data is supportive, is also likely to happen during the second and third quarters of next year.
Rezpeg incorporates a novel mechanism of action that has proven thus far to be safe and efficacious. It represents not just a single therapy but a building platform for multiple potential therapies addressing major multi-billion dollar markets.
There are a number of upcoming catalysts. In December, the topline results from the Phase IIb REZOLVE-AA study in patients with alopecia areata will be released, along with the outcome of the FDA end-of-Phase 2 meeting. In the first quarter of 2026, additional data from the atopic dermatitis Phase IIb REZOLVE-AD study will be released. Updates next year are also expected from the Phase 2 diabetes trial being conducted by TrialNet. Also, the Phase 3 start for AD and AA trials, if the data is supportive, is also likely to happen during the second and third quarters of next year.
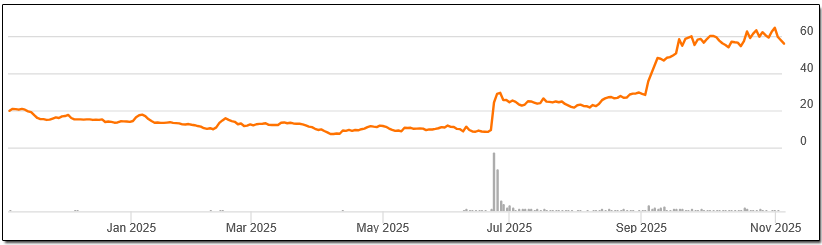
In our opinion, the company represents an attractive opportunity in the high-risk, high-reward biotechnology industry. We believe the stock price of Nektar Therapeutics can advance to $100 over the next 12 months, based on the success of its data releases. Nektar Therapeutics stock is already part of the Prudent Biotech model portfolio, which is up 44% this year as of October end, while the NYSE Biotechnology Index (IBB) is up 21%.
As additional data further derisks the program, and the promising potential of Rezpeg as a platform for additional treatments becomes more validated, it is likely that the company can invite acquisition interest. Last month, there was speculation of the same, though there was no official confirmation of such interest.
Positions and opinions are subject to change without notice. Investors should conduct their due diligence and recognize that biotechnology stocks carry elevated risk.
More details on the Phase 2b Study REZOLVE-AD study and pipeline details can be found in the corporate presentation.