Biotech Pulse
The biotech group closed 2019 with its longest rally of the year. It was a welcome change from what had mostly been an uphill battle. While the broader market was sailing from one high to another, biotechs and pharmaceuticals (biopharma) were still marking their 2019 lows in early October. But a confluence of events, including favorable political and regulatory outcomes, acted as a catalyst in pushing the indexes to a positive year after a decline in 2018. The Nasdaq Biotech Index (IBB) sharply rose for 2019 and so did the Prudent Biotech model portfolio.
We had expected 2019 returns to be in the 15% to 20% range, in the 2019 Outlook, and it was encouraging to see returns at the high end.
The year 2020 begins with the fundamentals for biotechs stronger than last year as the science continues to deliver, acquisitions have acquired a level of urgency, and there is plenty of risk capital to fund biotechs. But so has the risk of healthcare regulation become more prominent in the election year.
2020 Environment Remains Favorable
The macro-environment for stocks can be considered better than what we had in 2019.
The Sword of Damocles, the US-China trade friction, that precariously hung over the entire 2019, has been put back in its scabbard at least for now. This risk was so sharp in terms of stoking fear in the market that its dissipation will remove the most disruptive element for the stock market.
The trade deal should be a stimulus for the Chinese economy, diminish uncertainty for US agriculture and manufacturing along with alleviating tariff-related pricing pressures on US consumers, and provide a nudge to global trade.
US consumer spending, the mighty underpinning of US economic growth, remains strong, and the GDP is steady. The accommodative monetary policy, improving scientific advancements, and resurgent M&A are some other reasons that will help biotechs during the year.
Monetary Policy Spurs Biotech Risk-taking
In Summer 2019, we had noted that,
"A bullish scenario can be envisioned where the Federal Reserve cuts rates 2 to 3 times, in no small part due to the pressure from this trade conflict, which is then followed by a trade deal or a rollback, creating a highly favorable situation for the stock market." Trade War Has An Upside, August 2019
This is the bullish scenario that most likely will continue to persist in 2020.
After bringing down the interest rates, the bar to raise the level appears much higher for the Federal Reserve.
In order to move rates up, I would want to see inflation that's persistent and that's significant."
- Jerome Powell, Federal Reserve Chairman, at a press conference on 11 Dec 2019 after the FOMC meeting
This benign Federal Reserve rate outlook creates a favorable environment for biotech investing.
How?
In a stable economy, low-cost money attracts risk-taking.
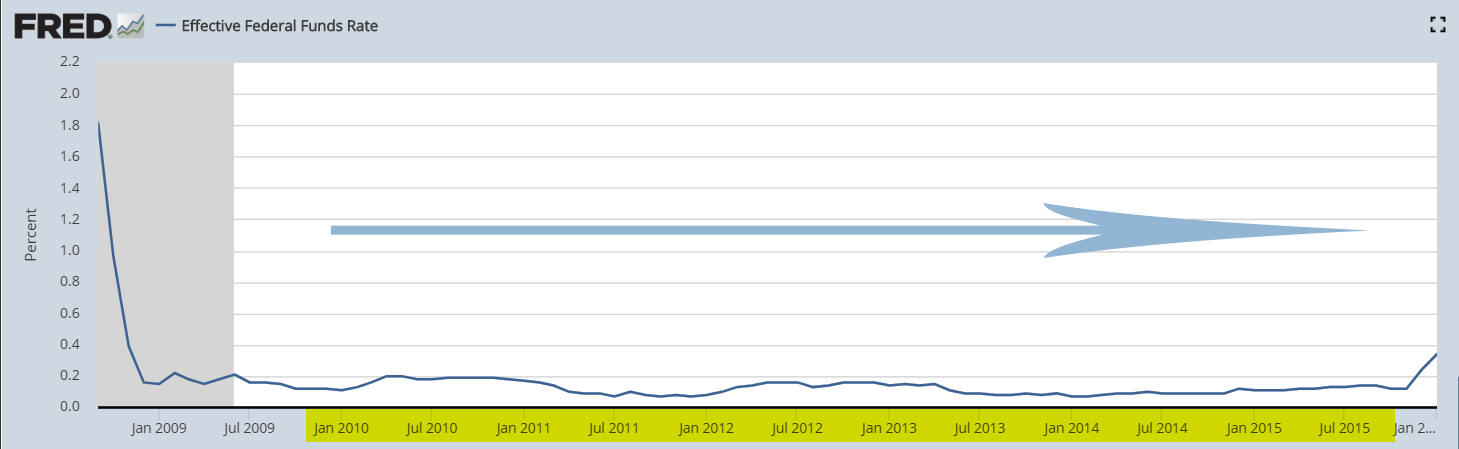
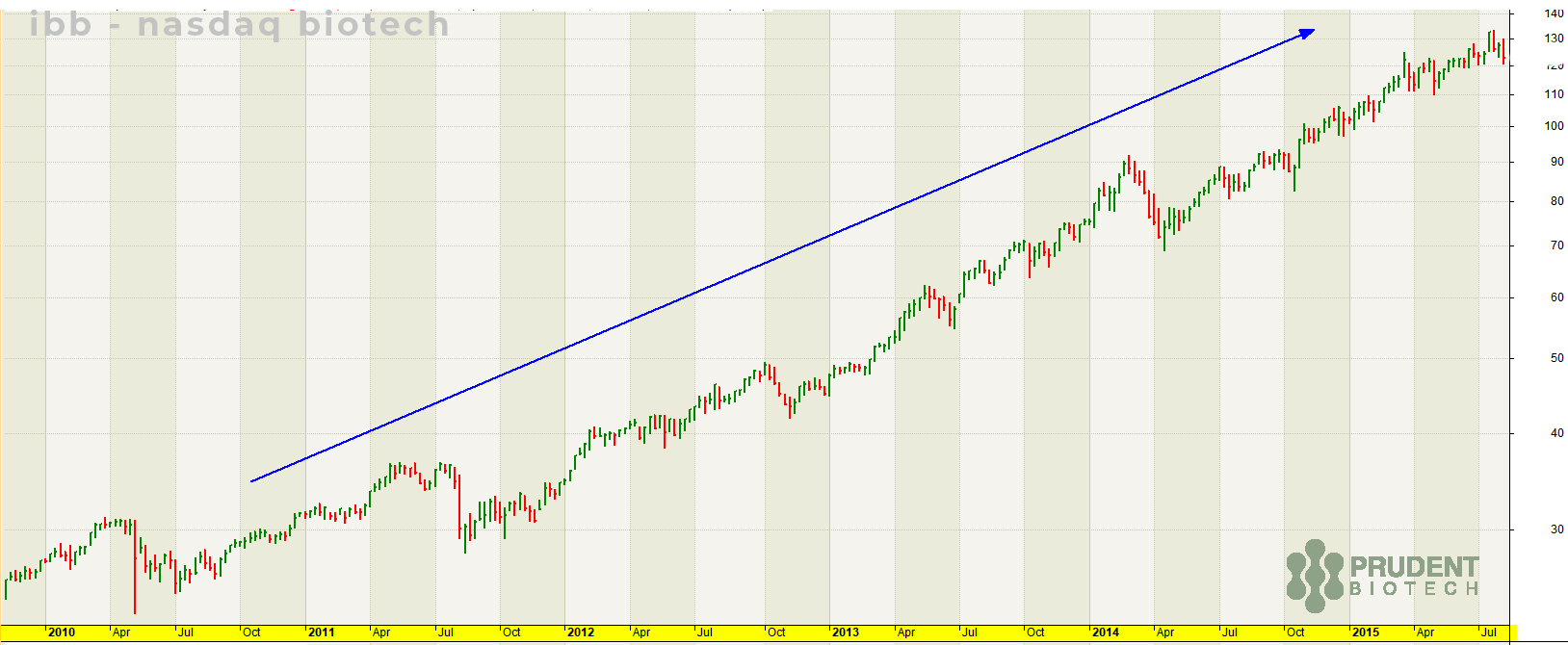
In the article, Biotechs Getting Ready To Explode, we had pointed to a correlation between low and stable rates versus biotech performance. To present this insight further, the best period during the past decade (2010-2019) for biotechs was the ~6-year period from 2010 to 2015 when the Nasdaq Biotech Index soared nearly 400%. This was a period of a stable interest rate policy after a reduction in interest rates.
When the Fed Funds rate (2010-2015) was flat after a downward bias...
...the biotech benchmark rose in a steady long-term uptrend from 2010 to 2015, recording its all-time high in July 2015.
It doesn't mean there is a perfect causal relationship between interest rates and biotech performance. But at the very least, the correlation underscores the existence of a favorable macro environment for biotechs that should continue in 2020.
Perhaps, biotech being the best industry performer in the fourth quarter last year is the beginning of a similar longer-term sustained rally.
Scientific Advances Keep Getting Better
The field of biotechnology is witnessing highly promising scientific advances uncovering innovative new cures for old maladies. And such advances keep getting better.
This decade should continue the age of biotechs as the human genome's basic building blocks are being further understood and harnessed to fight diseases. Breakthrough approaches and treatments are beginning to see the light of day. A few of them are mentioned below.
Gene therapy is providing a new platform to push forward cures for a wide variety of diseases that were considered incurable. It is a generational scientific advancement with the potential to make a gigantic impact on healthcare. We had discussed this topic in the article 3 Gene Therapy Stocks. The first gene editing treatment, Zolgensma, was approved by the FDA in 2019. The drug is owned by Novartis (NYSE:NVS) and is a treatment for spinal muscular atrophy. DNA editing is steadily advancing and overcoming obstacles to make delivering virus payloads more secure, safer (as it diminishes off-target effects), and potentially reversible (as the treatment intensity is managed).
RNAi drugs are finally beginning to gain traction after companies have been toiling in developing the space for the last two decades. Alnylam Pharmaceuticals (NASDAQ:ALNY) is leading the charge in RNA drug approvals. After its drug Patisiran became the first RNAi drug to be approved by the FDA in August 2018, Alnylam recently received a second approval for Givosiran in November 2019 and announced positive Phase 3 results in mid-December for Lumisiran. Another RNA drug, Inclisiran, for high cholesterol treatment, was also licensed from Alnylam, and The Medicines Company (NASDAQ:MDCO), which developed the drug, was acquired in late November after reporting positive Phase 3 results. The drug showed the effectiveness and the potential of RNA in resolving metabolic diseases. We had discussed the field of RNA in article 4 RNA Stocks For A Portfolio. Just like DNA editing, RNA is another approach in gene therapy that holds significant promise for investors and new variants of the approach are beginning to emerge.
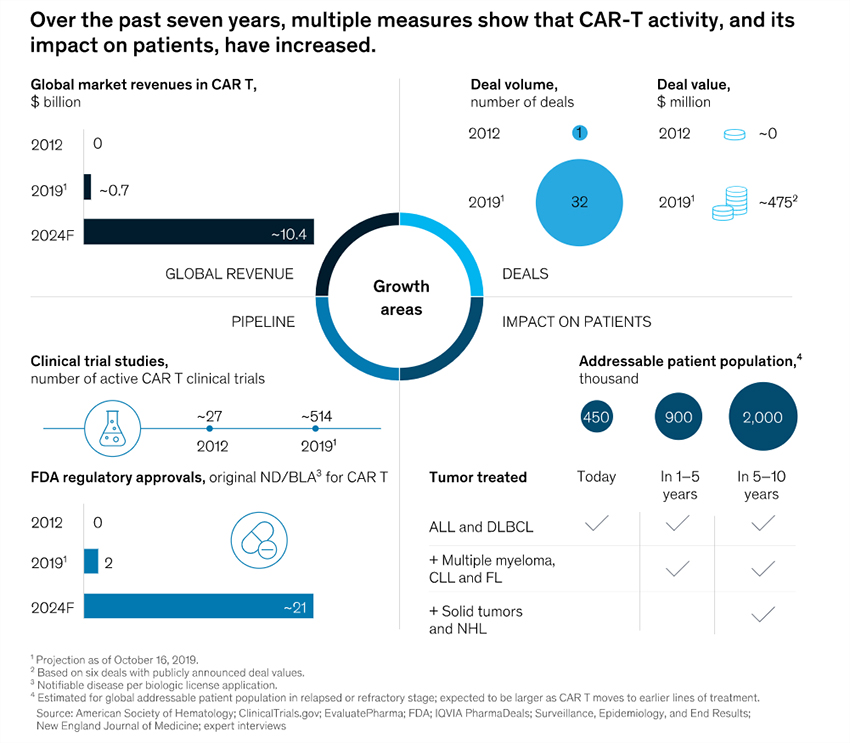
CAR T-cell therapies are disrupting cancer care for the better. They are proving themselves and are expanding into other targets and tumor types from CD19 targeting of earlier programs. The pipeline of investigative CAR T-cell therapies had grown to over 500 ongoing trials in 2019. This tide of trials is driven off the robustness of the clinical data which has proven the efficacy and effectiveness of the approach. The area is drawing billions in research budgets and will only keep getting more effective as the next generation of therapies will be easier to manufacture, expand to allogeneic therapies (cells from any healthy donor) from the more expensive autologous therapy (cells from the patient), and have better safety control triggers and mechanism to manage off-target effects. And the domain of CAR T-cell therapy will expand beyond just oncology. Scientists earlier in 2019 discovered a CAR T-cell therapy that shows complete resolution of the autoimmune disease Lupus in mice models. There are many clinical and commercial challenges with CAR-T, including those around manufacturing. But innovation will address them over time. Present CAR T-cell therapies address about 5% of the cancer patient market. This addressable population will expand and so will the market opportunity. Management consulting firm McKinsey forecasts CAR T-cell therapies to go from less than $1 billion global revenues in 2019 to over $10 billion in 2024.
A few months ago, Amgen (NASDAQ:AMGN) began Phase 2 trials of an investigational cancer drug that targets tumors caused by mutations in the KRAS gene. This is the first therapy that has been able to use the KRAS pathway. The KRAS genes are one of the most common causes of cancer due to errors in their crucial cell signaling protein. The mutated gene is believed to be present in around 90% of pancreatic cancers and 20% of non-small cell lung cancers. The KRAS oncogene has been resistant to therapies due to its smooth structure which makes it exceptionally difficult for a drug molecule to bind. Amgen made a breakthrough discovery of a pocket on the gene which can allow binding and has been able to drug the gene, potentially opening up a highly promising avenue for cancer therapy though we are still in early stages. Mirati Therapeutics (NASDAQ:MRTX) is also pursuing a similar approach, along with Eli Lilly (LLY) and Boehringer Ingelheim. But there appears to be a scarcity of KRAS - targeting companies, while the potential can turn out to be huge. Merck (MRK) this week agreed to offer $50 million upfront and up to $2.5 billion on milestones to license small-molecule inhibitors, including the KRAS oncogene, from Taiho and Astex.
Diseases of the central nervous system and mental disorders have been some of the most intriguing and complex diseases to resolve. Although the discipline of neurology was established 350 years ago, rapid progress has occurred in the last few decades as advances in technology provided the medium to further unravel the mysteries of the mind and the nervous system. But a lot remains unknown in this highly complex, interlinked, and broad field of neuroscience. The disorders are many and diverse, including prominent ones like Alzheimer's, schizophrenia, dementia, psychosis, epilepsy, insomnia, depression, Parkinson's, multiple sclerosis, etc.
After the beta-amyloid thesis was dismissed by one failed trial after another over decades, it was given a lifeline recently by Biogen's (BIIB) drug, Aducanumab, which achieved the rare distinction of both failing and then being claimed as a success in the space of six months. Aducanumab was resurrected last October and is being pitched by Biogen as ready for an FDA approval after a mild improvement in cognitive functions was noted in a subset of the population using a stronger dosage. The drug clears amyloid plaque from the brain. The beta-amyloid hypothesis states that the buildup of the protein beta-amyloid in the brain is central to the onset and progression of Alzheimer's. Thus, it contends that you wash away the plaque and you reverse the Alzheimer's symptoms.
We're far from solving the scourge of Alzheimer's and dementia and other related mental disorders. Most likely, the amyloid thesis has little traction left, and there is a clear yearning for new approaches.
Interestingly, a couple of weeks ago, Robert Moir, a Harvard scientist, who offered quite different, and often characterized as radical, theories in Alzheimer's, passed away. Dr. Moir's passing served as a reminder of his work that Alzheimer's needs to be tackled using different approaches. He postulated that the existence of beta-amyloid in the brain is for a reason, which is to defend it from infections. It's not the presence of the protein beta-amyloid that causes the disease, but most likely its over-production. Thus, clearing the brain of beta-amyloid can induce deleterious effects on the brain, and the research objective should be to manage its production. His work is no longer considered radical.
New approaches are being explored in CNS diseases and smaller biotechs are finding success. Karuna Therapeutics (KRTX) is pursuing a unique mechanism of action by relying on stimulation of muscarinic receptors rather than inhibiting dopamine receptors, as most therapies presently target, to reduce psychotic symptoms and cognitive impairment. Axsome Therapeutics (AXSM) and Intra-Cellular Therapies (ITCI) have found recent success with their programs. We had discussed neurology in a recent article.
The fusion of cheap money, large risk appetite, and rapid technological advances are shortening innovation cycles. The first-generation therapies are proving themselves and rapidly evolving into second-generation therapies.
Acquisitions & Partnerships
New frontiers of research and declining R&D productivity continue to raise pressure on the giant pharmaceutical companies.
After patiently waiting for a biotech acquisition surge for 2018, the acquisitions finally arrived in 2019 and picked up pace in the final quarter. The year started with the largest acquisition of the year with the purchase of Celgene by Bristol-Myers Squibb (BMY).
Major Biopharma Acquisitions of 2019
Celgene - acquired by Bristol-Myers Squibb (BMY) for $74 billion
Allergan - acquired by AbbVie (ABBV) for $63 billion
Array Biopharma - acquired by Pfizer (PFE) for $11.5 billion
Medicines Company - acquired by Novartis (NVS) for $9.7 billion
Loxo Oncology - acquired by Eli Lilly (ELY) for $8 billion
As science advances, it compels big pharmaceuticals to refresh their legacy pipeline, as well as seed it for the next generation of therapies. And science is advancing rapidly now with the big pharmaceuticals trying to catch up, leading to a bubbling transaction market.
Oncology remains one of the lead areas for acquisitions and partnerships. But there is a strong interest in targets across various areas, including rare diseases, gene therapy, RNA, ophthalmology, and neurology.
Most of the major pharmaceuticals and biotechs participated in acquisitions during 2019, including Novartis, Pfizer, Merck, Eli Lilly, Sanofi (SNY), Amgen, Vertex (VRTX), and Gilead (GILD). One would imagine Biogen would have to be seriously considering expanding its various therapeutic pathways in CNS-related diseases after significant doubts about the amyloid-beta thesis. Several companies have been mentioned frequently as potential targets, some for a long time. A few of these include CRISPR Therapeutics (CRSP), Sarepta Therapeutics (SRPT) and uniQure (QURE) in gene therapy; Amarin (AMRN) and Esperion Therapeutics in cardiovascular health and metabolic diseases; ACADIA Pharmaceuticals (ACAD) in neurology; Seattle Genetics (SGEN) in oncology; Global Blood Therapeutic for blood disorders; BioMarin Pharmaceuticals (BMRN) for rare diseases; and Incyte (INCY) for oncology and autoimmune.
While investing based on who gets acquired can be difficult and, often times, a futile exercise, an active transaction market raises the valuation for the entire sector. One good way to participate in the M&A theme is through a portfolio of promising mid-and-small-cap biotechs in attractive therapy areas, which can significantly appreciate in this environment even if they are not acquired. An acquisition or partnership would be a bonus.
Acquisitions will remain an ongoing theme in 2020 and convincing successes in Phase 1 and Phase 3 can invite buyer attention, particularly in much sought after therapeutic areas.
Noisier Regulatory Environment
Healthcare costs are the number one public interest issue.
Being a leading public issue, there is bipartisan support to address healthcare costs, although the paths being proposed by the two parties presently lead to opposite poles.
Nonetheless, the threat of greater regulation is an overarching risk with the potential to upend the industry dynamics.
And greater regulatory changes are coming to address healthcare costs.
The only question is the pace at which it happens.
Despite many industry victories, in the form of low wattage executive orders that did little damage to industry profitability, the threat of expanding regulation was a difficult issue in 2019 for investors. And, it will get even more difficult in 2020, an election year.
The House on 12 December 2019 passed a Democratic healthcare bill to control drug prices. The highlight of the bill is the ability of the Centers for Medicare and Medicaid to negotiate drug prices for at least 50 to up to 250 drugs. The bill will not progress in the Senate, but it does put pressure on the Republican-controlled Senate to come up with their drug price proposal. This being election year, both parties have to appear strong and resourceful on healthcare.
Drug price increases for 2020 have started. Over 200 drug prices were raised on January 1, as reported by Reuters. These single-digit price increases averaged around 6%. The price hikes will play into the intensifying political rhetoric as the first quarter proceeds.
Democratic Nomination
The biopharma industry and investors are so sensitive to the regulation risk that even the fortunes of candidates who are competing for the nomination can have a substantial impact on valuations based on the boldness of their healthcare plans. This was witnessed multiple times last year. The biopharma rally was triggered partly due to Warren slipping in the polls in October. But this is not over yet. Entering January, the candidate with the most momentum appears to be Bernie Sanders, and if he continues to close the gap with a more moderate frontrunner Joe Biden, then downside risk and volatility will return sharply to healthcare like it did last September.
The Democratic primary season is from February 3 to June 16. Most likely, the 3rd March Super Tuesday primary voting, when 14 states vote, and the 10th March elections, when 6 states vote, will clarify the race leader with the best chance for a Democratic nomination. If it turns out to be a candidate with a more moderate healthcare plan, like Biden, that should remove much uncertainty of a major change and be positive from a valuation standpoint. If candidates with more aggressive healthcare plans, like Warren and Sanders, are in the lead or still top competitors, then it would be a negative for biopharma valuations in March. A negative scenario can also be envisioned where a grand bargain between the Presidency and the House is reached along the lines of the recently passed House bill; however, the probability appears quite slim in the present environment.
Conclusion
The intrinsic fundamentals of the biotech sector have never been better. Even though the risk of a major regulatory shift will hound investors all year and will be the singular most important factor in determining the healthcare performance, in our opinion, biotechs couldn't be better placed fundamentally to face such an uncertain environment.
At this time, biotechs are on a trajectory that we believe will finally take the group, as represented by the Nasdaq Biotech Index, to an all-time high in the first-half and eclipsing the milestone recorded in July 2015. That would be quite a remarkable achievement, taking nearly 5 years. Such a long-term breakout typically augurs further gains over a long period. However, predicting the path of biotechs is hazardous in an election year where healthcare is amongst the top issues.
For the year, we anticipate the Nasdaq Biotech Index to rise 10%, with a much stronger first-half, held back by regulatory uncertainty in an election year. Many critical questions around regulation remain unanswered - the healthcare plan of the Democratic nominee, the makeup of Congress, etc. - which can measurably change the return expectation. Most likely, in the end, the country will end up with a stalemate in the Congress and White House on healthcare legislative priorities. If it becomes clearer that the industry structure will not change meaningfully, biotechs and pharmaceuticals can participate in a long and sustained rally.
More immediately, although it's time for a market consolidation after an uninterrupted run, and the first half of January can be rough, we believe the spike in acquisition activity and lower rates will continue to provide a scaffolding of strong support to biotech valuations for at least the first quarter. Also, the J. P. Morgan Healthcare conference, the largest industry event from Jan. 13 to 16, usually buoys the sector due to upbeat outlooks and transaction activity announced by management teams.
Biotech's fortunes will remain tied this year to scientific progress, and just as much to the outcome of the Presidential election and the impact on the industry structure. In a stock pickers market, there will be meaningful opportunities in biotech stocks to materially outperform the biotech indexes. But there will be times when reducing the exposure may be best.
The above return expectation is simply a framework providing general guidance and will need to be adjusted, up or down, as evolving conditions deviate meaningfully from current expectations. Biotechs being a high risk-reward industry, the risk management part is quite important to build returns over time. Investors need to pursue a concrete investment strategy in biotechs, preferring a portfolio approach by investing in a basket of promising biotech companies that can assist in managing risk and overcoming mistakes.
A few promising biotech companies, some of which may be now or in the past part of the Prudent Biotech or the Prudent Healthcare or Graycell Smallcap model portfolios, include Amgen (AMGN), Vertex Pharmaceuticals (VRTX), Alnylam Pharmaceuticals (ALNY), Seattle Genetics (SGEN), Arrowhead Pharmaceuticals (ARWR), Regeneron Pharmaceuticals (REGN), Mirati Therapeutics (MRTX), Reata Pharmaceuticals (RETA), BioMarin Pharmaceutical (BMRN), Global Blood Therapeutics (GBT), Sarepta Therapeutics (SRPT), ACADIA Pharmaceuticals (ACAD), Deciphera Pharmaceuticals (DCPH), Kodiak Sciences (KOD), CRISPR Therapeutics (CRSP), uniQure (QURE), Aimmune Therapeutics (AIMT), Amarin (AMRN), Assembly Biosciences (ASMB), Zymeworks (ZYME), Karyopharm Therapeutics (KPTI), Axsome Therapeutics (AXSM), Karuna Therapeutics (KRTX), Flexion Therapeutics (FLXN), Principia Biopharma (PRNB), Myovant Sciences (MYOV), Momenta Pharmaceuticals (MNTA), Apellis Pharmaceuticals (APLS), Arvinas (ARVN), Aurinia Pharmaceuticals (AUPH), Provention Bio (PRVB), and Iovance Biotherapeutics (IOVA). Industry exposure can also be acquired through ETFs like the one tracking the Nasdaq Biotech Index (IBB) and another tracking the S&P Biotechnology Select (XBI). In addition, there are higher-risk, leveraged ETFs for bullish (LABU) and bearish (LABD) outlooks.
Once again, take a portfolio approach in biotechs to overcome mistakes.
The article was first published on Seeking Alpha.